Lewis Dot Diagram Of Cl2
Lewis cl2 chlorine valence electrons Cl2 molecular orbital diagram Lewis atoms symbols diagram molecule structures covalent bonds chlorine structure cl bonding atom chemistry molecules two chem octet rule dot
Molecular Orbital Diagram For Cl2
Lewis dot diagram of co2 Mgcl2 lewis ionic magnesium molecular hybridization techiescientist polarity Correct lewis dot structure for chlorine (cl)?
Cl2 2nacl correct electron 2na molecule chlorine
2na(s) + cl2(g) → 2nacl(s) which of the following choices shows theMolecular orbital diagram for cl2 Molecular orbital diagram for cl2Molecular orbital diagram for cl2.
Cl2 lewis structureCl2 co2 fearless Dot lewis covalent bonds electron structures cl2 bond cl betweenLewis symbols and structures.

Chlorine dot
Lewis dot chlorine diagram structure cl electrons valence total cl2 draw thereMolecular orbital cl2 orbitals bonding delocalized atoms atomic energies chem libretexts Cl2 lewis structure ,valence electrons ,formal charge,polar or nonpolarOrbital molecular diagram cl2 s2 molecule mot molecules orbitals bond electron bonding unpaired draw c2 mo energy theory chlorine valence.
Mgcl2 lewis structure, molecular geometry, hybridization, and polarityMolecular orbital diagram diatomic molecules cl2 bond chemistry theory orbitals energy diagrams level bonding second delocalized homonuclear row electron h2 Covalent bonds in electron dot structures (lewis structuresMolecular orbital diagram oxide orbitals cl2 nitric diatomic mo energy level molecule principles molecules electrons delocalized theory bonding valence electron.


MgCl2 Lewis Structure, Molecular Geometry, Hybridization, and Polarity
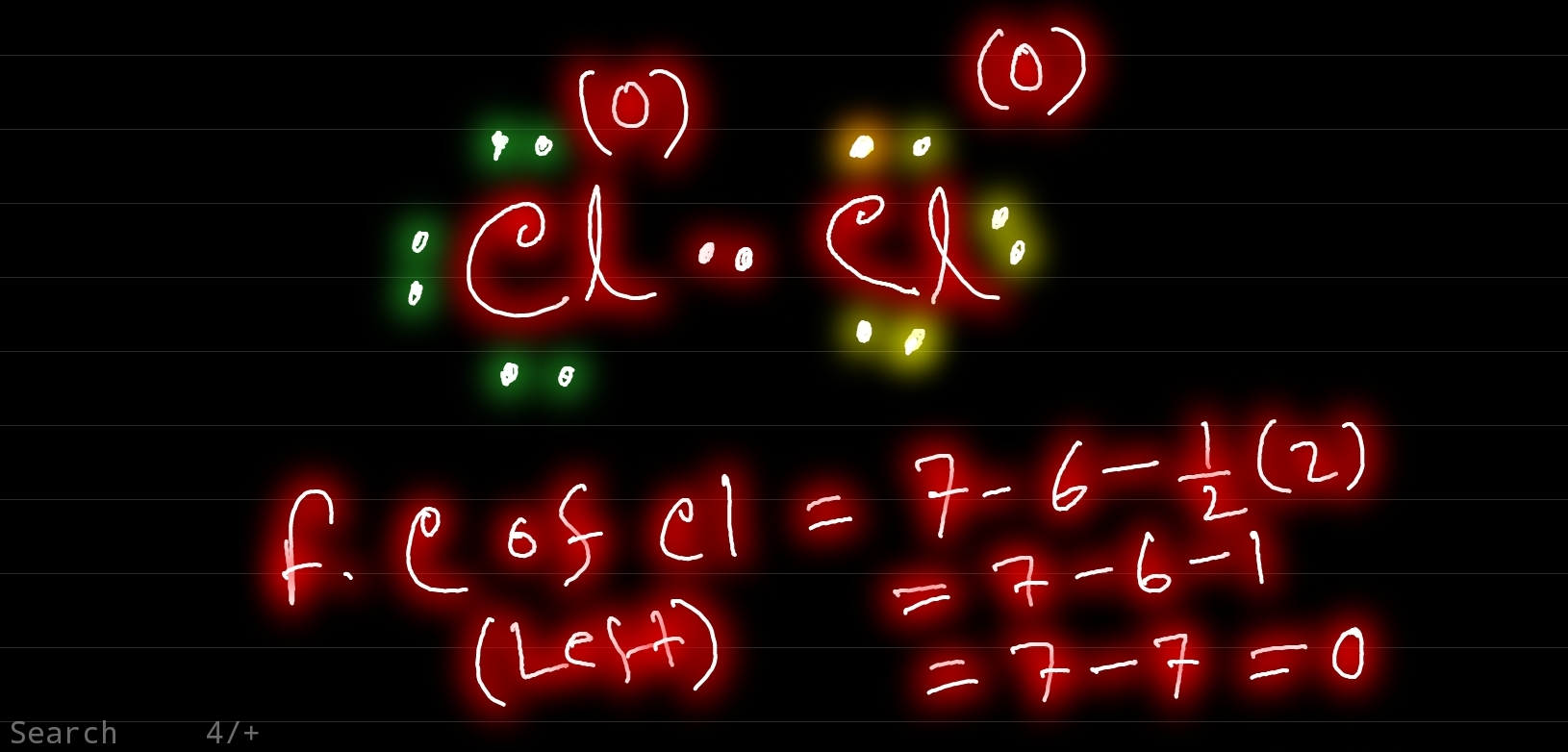
Cl2 Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar

Molecular Orbital Diagram For Cl2

correct lewis dot structure for chlorine (Cl)? - brainly.com

Molecular Orbital Diagram For Cl2

Lewis Symbols and Structures | Chemistry: Atoms First

Covalent Bonds in Electron Dot Structures (Lewis Structures

Cl2 Lewis Structure - How to Draw the Dot Structure for Cl2 - YouTube

2Na(s) + Cl2(g) → 2NaCl(s) Which of the following choices shows the

Lewis Dot Diagram Of Co2 - Drivenheisenberg